TL;DR A well-equipped research lab for mice/rats isn’t the one with the most gear—it’s the one with a stable, repeatable workflow. Prioritize anesthesia + scavenging, warming and temperature monitoring, physiological monitoring, ventilation when protocols demand it, non-invasive blood pressure (when needed), and a consistent surgical station with proper instrument cleaning/sterilization. Get those right and you protect animal welfare, technician safety, and data quality—without turning every study into a troubleshooting session.
What You Need for A Well-Equipped Research Lab
A practical guide to essential lab equipment for mouse and rat research workflows
A “well-equipped research lab” isn’t the one with the most gear.
It’s the one where teams can run the same procedure the same way—safely, consistently, and with stable physiology.
In practice, an equipped research lab is built around repeatable workflows that support animal welfare, technician safety, and reliable experimental data.
If you work with research animals, especially mice and rats, your equipment decisions have an outsized impact on the things that matter most: animal welfare, technician safety, workflow efficiency, and data quality. This guide breaks down the core equipment categories found in a preclinical research lab and explains what each is used for, when it becomes necessary, and what to prioritize.
Start with the workflow, not the shopping list
In a well-planned equipped research lab, these workflows form the operational backbone of daily rodent procedures:
- Anesthesia and recovery
- Temperature management
- Physiological monitoring
- Ventilation and intubation
- Blood pressure measurement
- Surgery and aseptic technique
- Training and repeatability tools
If you cover those well, you’ve covered the foundation of a capable mouse and rat research lab. These categories form the foundation of an equipped research lab designed for consistent rodent research workflows.
1) Anesthesia equipment for mice and rats
If your lab performs procedures that require sedation or surgery, anesthesia isn’t optional—it’s the gatekeeper for welfare and physiologic stability.
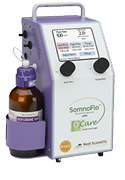
For small animal research, inhalant anesthesia is widely used because depth and duration are easier to control and recovery is typically faster than with many injectable agents. Inhalant anesthesia workflows often include:
- an anesthesia delivery system designed for rodents
- induction and maintenance components
- scavenging and safety accessories
- a repeatable recovery setup
The key is matching the system to the realities of mouse vs rat anesthesia: small tidal volumes, sensitivity to temperature drift, and the need for a stable, predictable workflow across staff.
Kent systems used in rodent anesthesia workflows include SomnoFlo®, SomnoFlo® O2Care, and SomnoSuite®.
What to prioritize
- stable control of anesthetic delivery
- repeatable setup that multiple technicians can run consistently
- clean transitions from induction → maintenance → recovery
2) Warming and temperature control
Temperature is one of the fastest ways rodent physiology drifts under anesthesia. Mice are especially prone to hypothermia because they lose heat quickly; rats have more thermal mass but still drift during longer procedures.
That’s why most well-equipped research labs treat warming and temperature monitoring as a standard part of the anesthesia workflow, not an extra.
A practical temperature setup usually includes:
- a reliable warming method during procedures
- temperature monitoring (core and/or surface) based on study needs
- a recovery station designed to maintain consistent conditions
The three most common warming approaches are warm water recirculating systems, electronic warming pads, or far-infrared warming systems. What matters most isn’t the technology label—it’s repeatability and control.
Kent warming tools include far-infrared warming pads, including the RightTemp® system, designed for consistent rodent support during anesthesia and recovery.
What to prioritize
- consistent warming during procedures
- a setup that prevents drift rather than reacting to it late
- repeatable conditions in the recovery area
3) Physiological monitoring during rodent procedures
A well-equipped research lab doesn’t monitor everything all the time. It monitors what protects the animal and interpretation of the study.
Common small animal physiological monitoring includes:
- heart rate
- oxygen saturation
- perfusion-related indicators
- respiration rate (directly or indirectly)
- temperature
For rodent anesthesia and recovery, pulse oximetry is often used to track oxygenation and heart rate trends—especially when procedures are long, anesthetic depth is variable, or stability is critical.
Kent monitoring systems commonly used in these workflows include both the MouseSTAT® Jr. and the PhysioSuite®, which integrates temperature management.
What to prioritize
- trend reliability during the procedure and recovery
- sensors and setups appropriate for mice vs rats
- monitoring that supports decisions, not just data collection
4) Ventilation and intubation equipment
Not every mouse or rat procedure requires mechanical ventilation. But when it does, it matters immediately.
Ventilation becomes more relevant when:
- procedures are longer
- anesthesia is deeper
- respiratory variables are part of the study
- recovery or oxygenation is inconsistent without support
- you are intubating and need controlled airflow
A small animal ventilator can provide more stable respiration, particularly in procedures where spontaneous breathing is unreliable or where you need consistency across animals.
Kent ventilation tools include RoVent® and RoVent® Jr., used in mouse and rat anesthesia workflows where controlled ventilation is part of the protocol.
What to prioritize
- repeatable respiratory support when needed
- weight-based setup that reduces operator variability
- compatibility with your anesthesia workflow and monitoring
5) Non-invasive blood pressure monitoring
Blood pressure is a high-value physiologic metric in many preclinical models. The challenge is measuring it in a way that fits the study design without unnecessary invasiveness.
Non-invasive tail-cuff systems are widely used for mouse and rat blood pressure monitoring because they support cohort work and avoid surgical implantation. Tail-cuff measurement is sensitive to stress, movement, and tail perfusion, so the workflow matters, but the method is well-established.
Kent’s CODA® non-invasive blood pressure monitor uses volume pressure recording technology to measure systolic, diastolic, and mean BP, along with heart rate and tail blood volume/flow in awake or anesthetized animals.
What to prioritize
- a stable workflow for acclimation and perfusion
- repeatable session design
- a method that supports multi-cohort studies
6) Surgical platforms, instruments, and aseptic workflow
When labs think “surgical tools,” they often focus on the instruments and forget the workspace. In rodent surgery, the station matters because it shapes consistency: where tools sit, how fluids are handled, how anesthesia lines are stabilized, and how warming is integrated.
A surgical platform can reduce variability by standardizing:
- instrument staging
- sterile boundaries
- fluid management
- animal positioning
- anesthesia tubing stabilization
- warming support
Kent’s SurgiSuite is designed as a multi-functional rodent surgical platform with an easy-to-clean base, integrated fluid channeling, and integrated warming, with mouse and rat sizing options.
On the instrument side, a well-equipped research lab typically maintains:
- procedure-appropriate scissors and forceps
- needle holders and suture-cutting tools
- retractors and probes
- duplicate sets for throughput
- a defined cleaning and sterilization process
Bead sterilizers can support rapid instrument tip sterilization in some workflows when allowed by SOP. They don’t replace cleaning and full sterilization, but they can help manage turnaround during high-throughput sessions.
What to prioritize
- consistency of the surgical workspace
- instruments that match field size and procedure type
- a sterilization workflow that preserves sharpness and alignment over time
7) Training tools that improve repeatability
Many labs also invest in tools that reduce variability by improving proficiency—especially for skills like tail vein injection or complex handling. Training tools aren’t just “nice.” They reduce rework, reduce stress, and improve consistency across operators.
Kent offers tail vein training devices that support skill development in a controlled way.
The simple way to think about a “well-equipped” research lab
A well-equipped research lab for mice and rats supports three outcomes:
- Stable animal physiology during procedures and recovery
- Repeatable workflows across people days
- Data you can trust without compensating with bigger sample sizes
If your lab covers anesthesia, warming, monitoring, ventilation (when needed), blood pressure, and a consistent surgical station, you’ve built a strong foundation.
Practical next step for your lab
If you tell us what you’re doing—mouse vs rat, procedure type, typical duration, awake vs anesthetized, and animals per session—we can help you map a workflow and equipment stack that fits your lab.
Well-equipped research lab FAQ
What equipment is needed in an equipped research lab with mice and rats?
Most rodent research labs need equipment for anesthesia, warming and temperature control, physiological monitoring, ventilation when required, non-invasive blood pressure measurement, surgical tools and a standardized surgical station, plus cleaning/sterilization and training tools that support repeatable techniques.
What is the most important equipment for rodent anesthesia?
A reliable rodent anesthesia delivery system, a safe scavenging setup, and a recovery workflow that includes warming and monitoring. Stability under anesthesia is usually determined as much by temperature and respiration as by anesthetic delivery.
Do mice and rats need different anesthesia equipment?
Often the same categories apply, but mouse vs rat work usually differs in sizing and tolerances. Mice drift faster and are less forgiving of setup variation, so mask fit, temperature control, and monitoring cadence tend to matter more.
What is the best way to prevent hypothermia in mice during surgery?
Use consistent warming during the procedure and recovery, and confirm temperature using an appropriate monitoring approach. Mice lose heat quickly under anesthesia, so treating warming as a standard step—rather than a rescue step—helps keep physiology stable.
What’s the difference between core and surface temperature monitoring in rodents?
Core temperature reflects true thermal status. Surface temperature changes quickly and is useful for trends and recovery triage, but it isn’t a reliable substitute for core. Many labs use both: core confirmation when it matters and surface checks for fast screening.
What physiological monitoring is commonly used in rodent surgery?
Common monitoring includes heart rate and oxygen saturation trends (pulse oximetry), temperature, and respiration trends. The right mix depends on procedure length, anesthetic depth, and whether physiologic stability affects the endpoint.
Do I need a ventilator for mouse anesthesia?
Not always. Ventilation becomes more relevant for longer procedures, deeper anesthesia, intubated workflows, or studies where respiratory control matters. For brief, light anesthesia many protocols rely on spontaneous breathing, but it should be a deliberate choice based on the procedure and endpoints.
What is a non-invasive blood pressure monitor for mice and rats?
Most non-invasive rodent BP monitoring uses a tail-cuff method that detects changes in tail blood volume/flow during cuff cycling. These systems are widely used for cohort studies because they avoid surgical implantation.
Is tail-cuff blood pressure accurate in mice and rats?
It can be reliable when the workflow supports it. Tail-cuff blood pressure measurement is sensitive to stress, movement, and tail perfusion, so acclimation, warming, and session design strongly influence repeatability.
What surgical instruments are essential for mouse and rat survival surgery?
A core set usually includes straight and curved dissecting scissors, fine atraumatic forceps, toothed forceps for skin, micro or mosquito hemostats, a needle holder, a dedicated suture cutter, retractors or exposure tools, and a defined cleaning/sterilization workflow. Mouse work often benefits from finer, micro-sized surgical instruments.
Why does a surgical platform matter in rodent surgery?
A standardized surgical station improves consistency by keeping instrument staging, sterile boundaries, anesthesia line management, fluid handling, and warming support predictable across users and across cases.
What categories of lab equipment are common even if not all suppliers carry them?
Beyond rodent procedure equipment, many labs also rely on caging/racks and husbandry systems, biosafety cabinets and sterilization equipment, pumps and infusion systems, imaging systems, behavioral/neuroscience tools, general analytical instruments, and EHS supplies like PPE and hazardous waste handling.
Common well-equipped research lab equipment most labs have
Facility and animal housing infrastructure
Most animal research programs rely on facility-level systems that are typically purchased through vivarium and cage-system vendors:
- ventilated caging and rack systems
- bedding and enrichment supplies
- water systems and bottle-fill stations
- cage washers and sanitation infrastructure
- environmental monitoring and logging for room conditions
Sterile processing and biosafety
Even when procedures happen outside a BSL workflow, many labs still depend on:
- biosafety cabinets or laminar flow hoods
- autoclaves and sterilizers
- instrument washers, ultrasonic cleaners, drying cabinets
- validated disinfectants, indicator strips, and sterile packaging supplies
General laboratory instruments and analytics
A well-equipped “research lab equipment list” usually includes core lab devices that are not animal-specific:
- balances and scales for reagents and compounds
- centrifuges, vortexers, shakers, and mixers
- incubators and cold storage (refrigerators, freezers)
- microscopes and imaging optics
- pipettes, consumables, tubes, plates, and sample storage systems
- spectrophotometers or plate readers depending on assays
Dosing, infusion, and fluid delivery systems
Many preclinical workflows require controlled delivery beyond basic injections:
- syringe pumps and infusion pumps
- osmotic pumps and accessories
- catheters, tubing, swivels, and tethering systems
- metabolic and fluid balance setups depending on model
Imaging systems and procedure-adjacent equipment
Depending on the model, labs may also use:
- ultrasound, micro-CT, MRI, PET, optical imaging
- procedure tables and imaging-compatible restraint accessories
- physiological monitoring optimized for imaging environments
Behavioral, neuroscience, and specialty research tools
Common categories in neuro/behavior programs include:
- stereotaxic systems and microinjection accessories
- behavioral mazes and tracking systems
- operant conditioning chambers
- electrophysiology and telemetry systems where required
EHS and compliance supplies
Often overlooked, always required:
- PPE and cleanroom supplies as applicable
- sharps containers and biomedical waste handling
- chemical hygiene and spill response supplies
- anesthesia scavenging compliance components and monitoring where required